Reviewed By Critical Care Assembly
Submitted by
Kriti Lonial, MD
Fellow
Division of Pulmonary, Critical Care and Sleep Medicine
University of Southern California, Keck School of Medicine
Los Angeles, CA
Pooja Patel, MD
Division of Pulmonary, Critical Care and Sleep Medicine
University of Southern California, Keck School of Medicine
Los Angeles, CA
A. Purush Rao, MD
Assistant Professor of Clinical Medicine
Division of Pulmonary, Critical Care and Sleep Medicine
University of Southern California, Keck School of Medicine
Los Angeles, CA
Submit your comments to the author(s).
History
A 38 year-old woman, with cystic fibrosis/bronchiectasis (genotype F508/F508) presented to the emergency department for sudden onset massive hemoptysis. She accounted an approximate volume of 6-8 ounces of blood. She denied any associated chest pain, worsened productive sputum or dyspnea prior to the massive hemoptysis episode. 2 hours later, she complained of a non-radiated, stabbing right-sided chest pain with an intensity of 3 out of 10 level.
Her past medical history included bronchiectasis, cystic fibrosis associated pancreatic insufficiency, anxiety and recurrent massive hemoptysis requiring prior bronchial artery embolization (BAE) up to 37 times during her lifetime. The most recent BAE prior to the current episode was 6 weeks prior.
She was a lifetime non-smoker, with no history of illicit drug or alcohol use.
Her medications included fat soluble vitamins (ADEK), pancreatic enzyme supplements, recombinant DNase and hypertonic saline. She denied any changes to her regimen prior to the most recent hemoptysis episode.
Physical Exam
A 38 year-old woman, with cystic fibrosis/bronchiectasis (genotype F508/F508) presented to the emergency department for sudden onset massive hemoptysis. She accounted an approximate volume of 6-8 ounces of blood. She denied any associated chest pain, worsened productive sputum or dyspnea prior to the massive hemoptysis episode. 2 hours later, she complained of a non-radiated, stabbing right-sided chest pain with an intensity of 3 out of 10 level.
Her past medical history included bronchiectasis, cystic fibrosis associated pancreatic insufficiency, anxiety and recurrent massive hemoptysis requiring prior bronchial artery embolization (BAE) up to 37 times during her lifetime. The most recent BAE prior to the current episode was 6 weeks prior.
She was a lifetime non-smoker, with no history of illicit drug or alcohol use.
Her medications included fat soluble vitamins (ADEK), pancreatic enzyme supplements, recombinant DNase and hypertonic saline. She denied any changes to her regimen prior to the most recent hemoptysis episode.
Lab
WBC: 6,200 per mm3
Hematocrit 37%
Platelet: 357,000 per mm3
PT/INR: 10.2/0.9
Electrolytes and liver function tests were normal
BUN was 10 mg/dL , and serum creatinine was 0.6 mg/dL
Sputum culture revealed mucoid pseudomonas aeruginosa, no burkholderia cepacia or staphloccus aureus
Sputum for AFB and fungi were negative
Figures

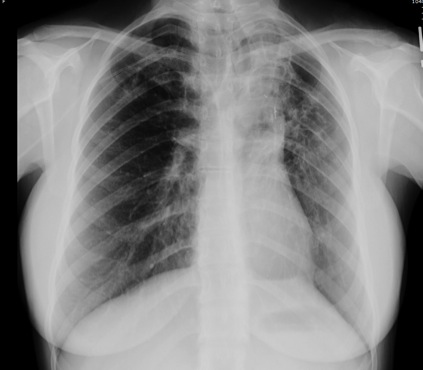
Figure 2: Chest computed tomography showing bronchiectasis and right upper lobe opacity suggesting the site of bleed
References
- Chaparro C, Maurer J, Gutierrez C. et al Infection with Burkholderia cepacia in cystic fibrosis: outcome following lung transplantation. Am J Respir Crit Care Med 2001. 16343–48.48.
- Aris R M, Routh J C, LiPuma J J. et al Lung transplantation for cystic fibrosis patients with Burkholderia cepacia complex. Survival linked to genomovar type. Am J Respir Crit Care Med 2001. 1642102–2106.2106.
- Courtney J M, Dunbar K E, McDowell A. et al Clinical outcome of Burkholderia cepacia complex infection in cystic fibrosis adults. J Cyst Fibros 2004. 393–98.98.
- De Soyza A, McDowell A, Archer L. et al Burkholderia cepacia complex genomovars and pulmonary transplantation outcomes in patients with cystic fibrosis. Lancet 2001. 3581780–1781.1781.
- Jones A M, Dodd M E, Govan J R. et al Burkholderia cenocepacia and Burkholderia multivorans: influence on survival in cystic fibrosis. Thorax 2004. 59948–951.951.
- Ledson M J, Walshaw M J. Burkholderia infection and survival in CF. Thorax 2005. 60439.
- Balfour‐Lynn I, Walters S, Dezateux C. Inhaled corticosteroids for cystic fibrosis. Cochrane Database Syst Rev 2000. (1)
- Bradley J M, Moran F M, Elborn J S. Evidence for physical therapies (airway clearance and physical training) in cystic fibrosis: an overview of five Cochrane systematic reviews. Respir Med 2006. 100191–201.201.
- Elkins M R, Jones A, van der Schans C. Positive expiratory pressure physiotherapy for airway clearance in people with cystic fibrosis. Cochrane Database Syst Rev 2006. (2)
- Lands L C, Dezateux C, Crighton C. Oral non‐steroidal anti‐inflammatory drug therapy for cystic fibrosis. Cochrane Database Syst Rev 1999.
- Main E, Prasad A, van der Schans C. Conventional chest physiotherapy compared to other airway clearance techniques for cystic fibrosis. Cochrane Database Syst Rev 2005.
- Cheng K, Ashby D, Smyth R. Oral steroids for cystic fibrosis. Cochrane Database Syst Rev 1999. (4)
- Smyth A, Walters S. Prophylactic anti‐staphylococcal antibiotics for cystic fibrosis. Cochrane Database Syst Rev 2003. (3)
- Southern K W, Barker P M, Solis A. Macrolide antibiotics for cystic fibrosis. Cochrane Database Syst Rev 2004. (2)
- van der Schans C, Prasad A, Main E. Chest physiotherapy compared to no chest physiotherapy for cystic fibrosis. Cochrane Database Syst Rev 2000. (2)
- Goss C H, Burns J L. Exacerbations in cystic fibrosis: epidemiology and pathogenesis. Thorax 2007. 62360–367.367.
- Duijvestijn Y C, Brand P L. Systematic review of N‐acetylcysteine in cystic fibrosis. Acta Paediatr 1999. 8838–41.41.
- Fuchs H J, Borowitz D S, Christiansen D H. et al Effect of aerosolized recombinant human DNase on exacerbations of respiratory symptoms and on pulmonary function in patients with cystic fibrosis. The Pulmozyme Study Group. N Engl J Med 1994. 331637–642.642.
- Quan J M, Tiddens H A, Sy J P. et al A two‐year randomized, placebo‐controlled trial of dornase alfa in young patients with cystic fibrosis with mild lung function abnormalities. J Pediatr 2001. 139813–820.820.
- McCoy K, Hamilton S, Johnson C. Effects of 12 weeks administration of dornase alfa in patients with advanced cystic fibrosis lung disease. Chest 1996. 110889–895.895.
- Robinson M, Regnis J A, Bailey D L. et al Effect of hypertonic saline, amiloride, and cough on mucociliary clearance in patients with cystic fibrosis. Am J Respir Crit Care Med 1996. 1531503–1509.1509.
- Wark P A B, McDonald V, Jones A P. Nebulised hypertonic saline for cystic fibrosis. Cochrane Database Syst Rev 2005. (3)
- Suri R, Metcalfe C, Lees B. et al Comparison of hypertonic saline and alternate‐day or daily recombinant human deoxyribonuclease in children with cystic fibrosis: a randomised trial. Lancet 2001. 3581316–1321.1321.
- Elkins M R, Robinson M, Rose B R. et al A controlled trial of long‐term inhaled hypertonic saline in patients with cystic fibrosis. N Engl J Med 2006. 354229–240.240.
- Homnick D N, White F, de Castro C. Comparison of effects of an intrapulmonary percussive ventilator to standard aerosol and chest physiotherapy in treatment of cystic fibrosis. Pediatr Pulmonol 1995. 2050–55.55.
- Do ̈ring G, Flume P, Heijerman H, Elborn JS, Consensus Study Group. 2012. Treatment of lung infection in patients with cystic fibrosis: Current and future strategies. J Cyst Fibrosis 2012 11: 461–479.
- Balough K, McCubbin M, Weinberger M, Smits W, Ahrens R, Fick R. 1995. The relationship between infection and inflammation in the early stages of lung disease from cystic fibrosis. Pediatr Pulmonol 20: 63–70.
- Gibson R L, Emerson J, McNamara S. et al A randomized, controlled trial of inhaled tobramycin in young children with cystic fibrosis (abstract). Pediatr Pulmonol 2002. 34(Suppl 24)300.
- Valerius N H, Koch C, Hoiby N. Prevention of chronic Pseudomonas aeruginosa colonisation in cystic fibrosis by early treatment. Lancet 1991. 338725–726.726.
- Ratjen F, Doring G, Nikolaizik W H. Effect of inhaled tobramycin on early Pseudomonas aeruginosa colonisation in patients with cystic fibrosis. Lancet 2001. 358983–984.984.
- Wiesemann H G, Steinkamp G, Ratjen F. et al Placebo‐controlled, double‐blind, randomized study of aerosolized tobramycin for early treatment of Pseudomonas aeruginosa colonization in cystic fibrosis. Pediatr Pulmonol 1998. 2588–92.92.
- Ramsey B W, Pepe M S, Quan J M. et al Intermittent administration of inhaled tobramycin in patients with cystic fibrosis. Cystic Fibrosis Inhaled Tobramycin Study Group. N Engl J Med 1999. 34023–30.30.
- Saiman L, Marshall B C, Mayer‐Hamblett N. et al Azithromycin in patients with cystic fibrosis chronically infected with Pseudomonas aeruginosa: a randomized controlled trial. JAMA 2003.
- Wolter J, Seeney S, Bell S. et al Effect of long term treatment with azithromycin on disease parameters in cystic fibrosis: a randomised trial. Thorax 2002. 57212–216.216.
- Equi A, Balfour‐Lynn I M, Bush A. et al Long term azithromycin in children with cystic fibrosis: a randomised, placebo‐controlled crossover trial. Lancet 2002. 360978–984.984.
- Szaff M, Hoiby N, Flensborg E W. Frequent antibiotic therapy improves survival of cystic fibrosis patients with chronic Pseudomonas aeruginosa infection. Acta Paediatr Scand 1983. 72651–657.657.
- Hoiby N, Koch C. Cystic fibrosis. 1. Pseudomonas aeruginosa infection in cystic fibrosis and its management. Thorax 1990. 45881–884.884.
- Davies J, Sheridan H et al. Assessment of clinical response to ivacaftor with lung clearance index in cystic fibrosis patients with a G551D-CFTR mutation and preserved spirometry: a randomised controlled trial. Lancet Respir Med. 2013.; 1(8):630-8.
- Davies JC, Wainwright CE et al. Efficacy and safety of ivacaftor in patients aged 6 to 11 years with cystic fibrosis with a G551D mutation. Am J Respir Crit Care Med. 2013; 187(11): 1219-25.
- Ramsey BW, Davies J, et al. A CFTR potentiator in patients with cystic fibrosis and the G551D mutation. NEJM, 2011;365(18):1663-72.
- Accurso FJ, Rowe SM et al. Effect of VX-770 in persons with cystic fibrosis and the G551D-CFTR mutation. NEJM 2010; 363(21): 1991-2003.
- Flume PA, Liou TG et al. Ivacaftor in subjects with cystic fibrosis who are homozygous for the F508del-CFTR mutation. Chest 2012; 142(3):718-24.
- Rogan MP, Stoltz DA, Hornick DB. Cystic fibrosis transmembrane conductance regulator intracellular processing, trafficking, and opportunities for mutation-specific treatment. Chest 2011; 139: 1480–1490.
- De Boeck K, Kent L, Davies J, et al. CFTR biomarkers: time for promotion to surrogate end-point. Eur Respir J 2013; 41: 203–216.
- Elborn JS. Personalised medicine for cystic fibrosis: treating the basic defect. Eur Respir Rev 2013; 22: 3–5.
- Derichs N. Targeting a genetic defect: cystic fibrosis transmembrane conductance regulator modulators in cystic fibrosis. Eur Respir Rev 2013; 22: 58–65.
- Yu H, Burton B, Huang CJ, et al. Ivacaftor potentiation of multiple CFTR channels with gating mutations. J Cyst Fibros 2012; 11: 237–245.